How the body ages is one of the oldest questions in medicine. Yesterday, for the first time, we got something close to an answer.
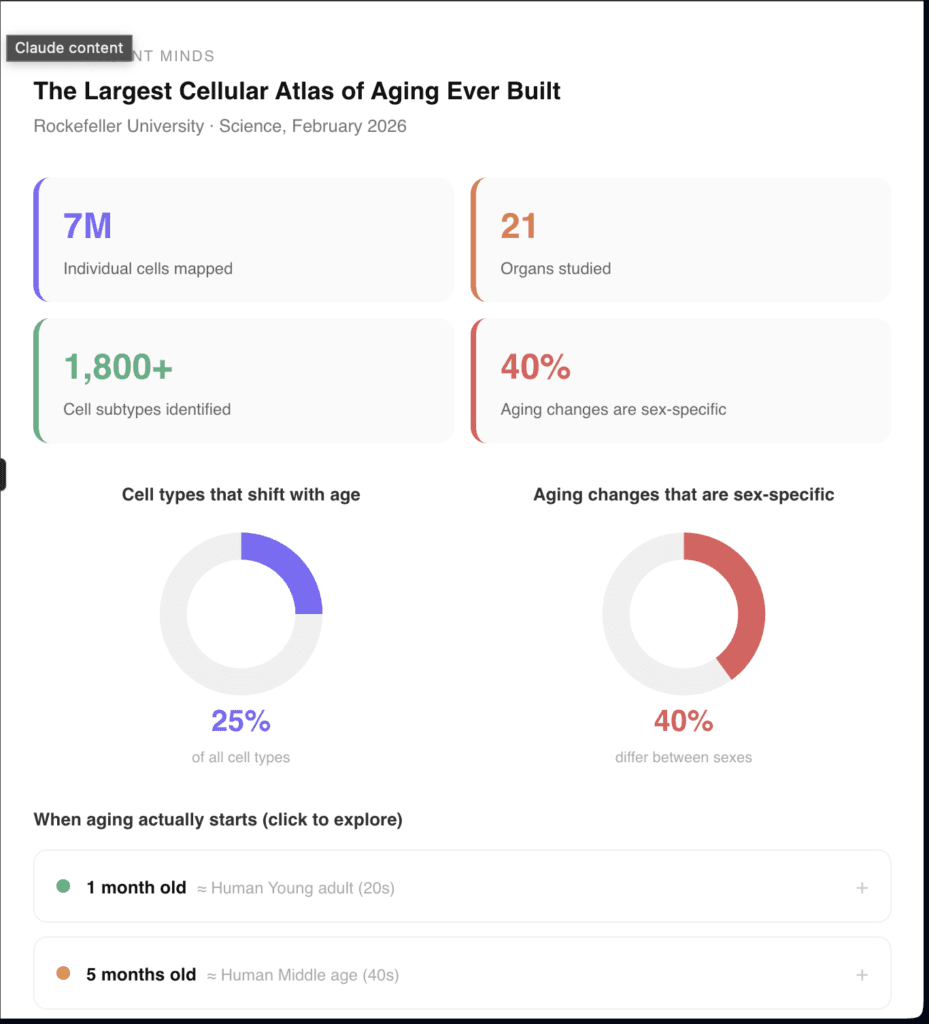
A study published in Science on February 26, 2026, by researchers at The Rockefeller University mapped nearly 7 million individual cells across 21 different organs, tracking exactly what happens to each cell type as the body moves from young adulthood through middle age into old age. It is the most comprehensive cellular atlas of aging ever built. And what it found overturns several assumptions that have guided biology for decades.
Aging is not random. It is not each organ quietly deteriorating on its own schedule. It is coordinated, systematic, and in some ways almost orchestrated.
What Was Actually Built
The team, led by graduate student Ziyu Lu under professor Junyue Cao, used a technique called single-cell ATAC-seq to examine how DNA is packaged inside each individual cell. When DNA is open and readable in a particular region, the genes in that area can become active. When it is tightly packed, they stay silent. By mapping which regions were open and which were closed across millions of cells, the researchers could see not just which genes were changing but which genetic switches were being rewired.
They studied 32 mice at three life stages: one month old, which corresponds to young adulthood; five months, which corresponds to middle age; and 21 months, which corresponds to old age.
In total they identified more than 1,800 distinct subtypes of cells, including many rare populations that had never been characterised before. They then tracked exactly how the numbers of each subtype shifted across the three age groups.
Scientists once believed that aging mainly altered how cells functioned, not how many of each type were present. This study challenges that assumption directly.
What They Found
Roughly one quarter of all cell types across the 21 organs showed significant shifts in their populations with age. Some types expanded. Others contracted. Some disappeared almost entirely in old age. Others appeared for the first time.
Immune cells expanded broadly. Specialised cells in the kidney, muscle and lung declined. The pattern was not uniform across the body but it was not random either.
The most unexpected finding was the coordination.
The same cellular changes appeared and declined in parallel across completely different organs. A particular immune cell subtype would rise simultaneously in the liver, the lung and the spleen.
A specialised structural cell would decline at the same rate in tissues that have no direct biological connection to each other.
This synchronisation suggests that aging is not driven organ by organ. It is driven by body-wide signals. The most likely candidate is cytokines, immune signalling molecules that circulate in the bloodstream and can send instructions to multiple organs at once.
When the researchers compared their data with previous studies, they found that cytokines could trigger many of the same cellular changes they observed in aging mice.
Junyue Cao described it this way: our goal was to understand not just what changes with aging, but why. By mapping both cellular and molecular changes, we can identify what drives aging. That opens the door to interventions that target the aging process itself.

The Sex Difference Nobody Expected
About 40 percent of all aging-associated cellular changes in the study were significantly different between males and females.
That number is striking. It means that nearly half of what aging does to the body depends on whether you are male or female. The same chronological age produces different biological outcomes in different bodies.
The most pronounced difference involved immune activation. Females showed much broader immune activation with age than males.
Cao speculated that this could explain the higher prevalence of autoimmune diseases in women, a pattern that has been observed clinically for decades but never understood mechanistically.
Males, by contrast, showed patterns consistent with higher risk of conditions like Parkinson’s disease in later life.
The implication for medicine is significant. Most aging research and most drug development has historically used male subjects, male cell lines and male animal models.
If 40 percent of aging-related changes are sex-specific, then interventions developed in male models may produce very different results in female bodies.

Where Aging Actually Starts
One of the study’s most practically important findings was that many age-related changes begin earlier than expected.
The shift from young adulthood to middle age in the mouse model, equivalent to the transition from early adulthood to middle age in humans, showed measurable cellular changes in multiple organ systems. Not dramatic changes. But directional ones, pointing clearly toward the patterns that would become pronounced in old age.
By the time the visible signs of aging appear, a great deal has already happened at the cellular level.
This reframes where anti-aging medicine should be focused. If the biological changes start in middle age, or earlier, waiting until age-related disease is already present may mean intervening too late. The window for meaningful prevention is wider than previously assumed, but it opens earlier.
The researchers identified specific genomic hotspots, regions where DNA accessibility changes were consistent across multiple cell types during aging, that could become targets for therapeutic intervention.
These hotspots are not random. They cluster around regulatory regions that control immune function and cellular stress responses. Drugs that modulate cytokine signalling, Cao suggested, could potentially slow the coordinated aging processes across many organs simultaneously.

The Graduate Student Who Built the Map
There is a detail in this study worth noting.
The entire atlas, 7 million cells across 21 organs, the most comprehensive map of aging ever assembled, was built by a single graduate student.
Most projects of this scale require large international consortia, dozens of laboratories, and years of coordinated effort. This one was done by Ziyu Lu, using a method that Cao’s lab optimised specifically to make the process efficient enough for a single researcher to manage.
Cao described it as remarkable. It is also a signal about what becomes possible when the methods are right.
The full atlas is now publicly available at epiage.net. Any researcher in the world can access it, search it by organ, by cell type, by age group, by sex. It is a shared infrastructure for everyone working on aging, offered freely.
What Comes Next
Cao was precise about where the research currently stands. This is really a starting point. We have identified the vulnerable cell types and molecular hotspots. Now the question is whether we can develop interventions that target these specific aging processes. Our lab is already working on that next step.
The atlas does not offer an anti-aging therapy. It does not reverse anything. What it provides is a detailed, high-resolution map of the territory, the first honest answer to the question of what is actually happening inside the body as it ages.
Every intervention that gets developed from here will be developed with this map as its foundation. Every drug trial, every therapeutic hypothesis, every biomarker that researchers want to track will be tested against what this atlas describes.
This study will not be the thing that extends human healthspan. But it may be the thing that makes the thing that extends human healthspan possible.
That is what foundational science looks like. Not a cure. A map.
Read next: The world as we knew of it, as of yesterday, is no more
What marketing actually is (it is not what most people think)
Tagore’s The Postmaster: a story about being left behind
What is dharma? what the mahabharata knew about losing everything




